AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic mass number of calcium11/18/2023 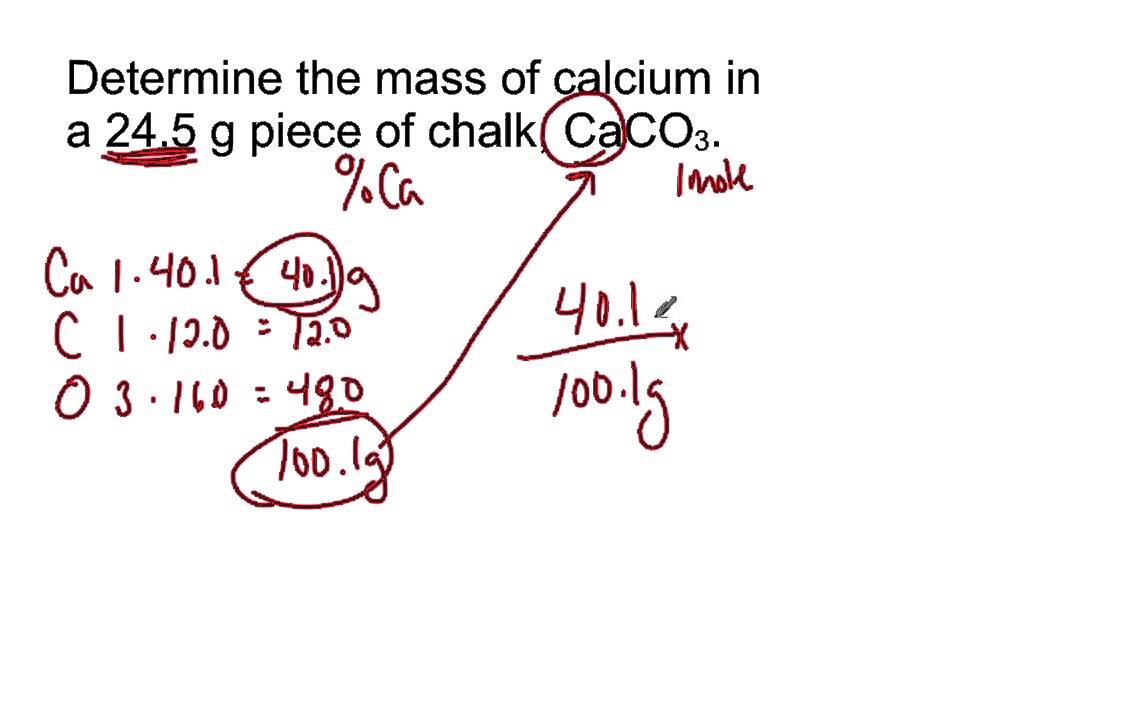
There is one more positive charge than negative charge : +1ģs (max = 2), 3p (max = 6), 3d (max = 10) Total = 18 There is one more negative charge than positive charge : -1 If one electron is removed from a sodium atom The outer most electron(s) are termed the valence electrons. Two electronsĪre in the K shell, 8 in the L shell and one in the M shell. Actually, there are sub levels within the main level (except for theįor element 11 (Sodium - Na) there are 11 electrons and 11 protons. 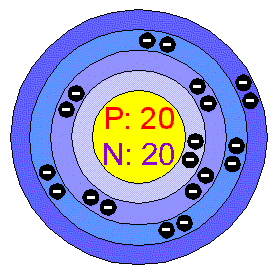
Second electron goes into the first level (atomic Helium) and the Start with a single electron - it occupies the empty There are 20 electrons surrounding is nucleusĪ useful exercise is to start with the first element (Hydrogen) with its one proton and one electron and add protons and electrons to build up the remaining chemical elements. Number of positive particles (electrons = protons).Ī atom of calcium has 20 neutrons. In a neutral atom the number of negative particles equals the The maximum occupancy = 2n 2 where n is the level. The first level (K) is closest to the nucleus.Īs the principal number increases 1.2.3.4 the number of electrons that can reside in the level increases. The primary division of these levels is called the principal quantum number. Electrons are "attracted" to the nucleus (positive and negative charges attract). In fact, much of the volume of an atom consists of empty space. Recognition that the distribution of electrons around the nucleus is not random was one of the great accomplishments of physicists in the 1920s - quantum mechanics.Įlectrons occupy "levels" which are separated from each by some volume of space which electrons can pass through but not remain. In addition to the particles in the nucleus, there are negatively charged particles - electrons which "orbit" the central nucleus. There are 20 protons and 42 neutrons in its nucleus There are 20 neutrons and 22 protons in its nucleus There are 20 protons and 22 neutrons in its nucleus (How many neutrons does each isotope possess?)Ī calcium atom is found with a mass of 42. Atoms which have the same atomic number but different mass numbers are isotopes.įor example, all calcium atoms have an atomic number of 20 but there are three isotopes of calcium - 39, 40, and 42. Neutrons in their nucleus as all must have the same number of protons - 20. If we could weigh individual atoms of calcium we would find some variation in their mass this must be due to variations in the number of There are 20 protons in the nucleus of a Calcium atomĪ Calcium atom weighs 20 atomic mass units There are 20 neutrons in the nucleus of a Calcium atom You should learn the symbols for the eight most abundant elements in the Earth's crust. Each element is assigned a symbol - H for hydrogen. All atoms of an element have the same number of protons - hence the same atomic number for example all atoms of hydrogen have one proton in the nucleus. The atomic mass number is the number of protons plus neutrons in the nucleus. The atomic number is the number of protons in the nucleus of the atom. protons which carry a positive charge andįor our purposes we will ignore quarks and other particles that we would encounter if this were a upper level physics course.These spheres contain a central nucleus which contains two types of particles: What happened to the "missing 2" will be left for you to think about. There are 92 so-called naturally occurring elements of which 90 can be detected on Earth at this time. The radii of these atoms are a few angstroms (where an angstrom is 10 -10 cm. A rather crude, but useful, picture of an atom is that of a rigid sphere. has a fixed chemical composition or a composition that varies over a known extentĪll matter is made up of chemical elements, each of which is made up of particles called atoms.is inorganic - not formed as part of a life process.Gas - flows easily and expands to fill its container - disordered structure.Liquid - flows and conforms to the shape of its container - short range order.Non-Crystalline Solid - as above but the structure exhibits a short range order - glass is an non crystalline or amorphous solid.Crystalline Solid - as above and the structure (distribution of the components) is highly regular and repetitive - long range order.Solid - rigid substance that retains its shape unless distorted by a force.The following classification will help define the states in which matter can occur. The Earth - Introduction Class Notes - MineralsĪnything that has mass and occupies space is matter.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
